True navigation is defined as the
ability of an animal to return to its original location after
displacement to a site in unfamiliar territory, without access to
familiar landmarks, goal emanating cues, or information about the
displacement route. This ability, which has been demonstrated in
vertebrates (Rodda & Phillips, 1992) and one invertebrate (spiny
lobster; Boles & Lohmann, 2003), requires both a sense of direction
(ōcompassö), and a sense of geographic position (ōmapö).
Vertebrates
have multiple compass systems (sun, star, polarized light and magnetic
compasses; Able, 1980; Emlen, 1975; Lohmann & Lohmann, 1996; Moore, 1987; Phillips, 1986b;
Schmidt-Koenig, 1979; Wiltschko & Wiltschko, 1972; Wiltschko & Wiltschko,
1995a; see also Bingman et al.,
this volume). Factors that determine which of these compass systems
is used at any given time include weather conditions, time of day,
and past experience. Each of these compass systems requires
different sensory detection/processing mechanisms, e.g., a time
compensation mechanism for the sun compass (Phillips & Waldvogel,
1988; Saint Paul, 1953; Schmidt-Koenig, 1990), an ability to relate
the alignments of star patterns to the axis of celestial rotation
for the star compass (Emlen, 1970; Sauer & Sauer, 1960; Weindler,
Wiltschko, & Wiltschko, 1996; Wiltschko, Daum, Fergenbauer-Kimmel, &
Wiltschko, 1987), and specialized sensory receptors
capable of detecting the plane of polarized light and alignment of the
geomagnetic field for the polarized light and magnetic compasses
(Able & Able, 1993; Brines & Gould 1982; Phillips & Moore, 1992). Each compass system also incorporates to varying degrees
both innate and learned components (Able & Able, 1997; Bletz, Weindler,
Wiltschko, & Wiltschko, 1996; Gwinner & Wiltschko, 1978;
Helbig, 1992, 1996). To avoid systematic errors in the direction of orientation
when switching between compasses, each of these systems must be
calibrated with respect to a common reference system. In birds,
where the integration of compass information is best understood, the
primary compass calibration reference appears to be derived from
celestial cues, probably polarized patterns present at sunset and,
possibly, also sunrise (Muheim, Moore, & Phillips, 2006a; Muheim, Phillips, &
Akesson, 2006b).
Accurate
navigation only requires that the map and compass are in register
with one another, i.e., that the animal navigator is able to
associate a geographic position specified by the map with a compass
bearing that will enable it to return to the origin of a
displacement or to some other predetermined destination. As noted
above, however, birds appear to use a global reference system
derived from celestial polarized light patterns to calibrate their
compass and presumably, therefore, also their map systems.
The key feature
of a navigational map is that it can be extrapolated to unfamiliar
sites and used to orient homeward when there is no contact with
familiar ōlandmarksö or reference points, or with goal-emanating
cues. This definition of a navigational map applies to both
olfactory and magnetic gradient maps (see below), but not olfactory
mosaic maps (Able, 2000; Gould, 1982; Griffin, 1952; Wehner, 1998) or
other forms of ōpilotingö or ōplace navigationö involving the use of
familiar visual, olfactory or magnetic reference points. A
ōgradientö map requires an animal to learn the alignment and,
possibly, steepness of one or more environmental gradients within
its home range or territory, and to extrapolate these gradient(s)
beyond its area of familiarity. Comparison of the value of such a
map component at an unfamiliar site with that of the ōhome valueö
provides information about the animalÆs position along the gradient
in relationship to home. Non-parallel gradients of two or more
different map components would enable an animal to determine its
position in two dimensions (bicoordinate position fixing).
The home value of
each map component provides the reference necessary to determine the
direction (and distance) of displacement along each extrapolated
gradient. Since both magnetic and olfactory gradients change over
time (Courtillot, Hulot, Alexandrescu, le Mouļl, & Kirschvink, 1997; Ganzhorn, 1995; Skiles, 1985; Waldvogel, 1987; and see below), the home value(s) of an olfactory or magnetic
map would have to be updated periodically. The animal, therefore,
must have a means of distinguishing home from non-home locations
(ōhome recognitionö cues) that is independent of the cues used to
derive geographic position (ōmapö cues). Manipulation of home
recognition cues may produce dramatic changes in homing orientation
and/or homing success that can easily be mistaken for an effect on
cues underlying the map (see below).
A unified theory
of spatial navigation requires integration of mechanisms used to
guide movements within familiar configurations of landmarks
(piloting, place maps, bearing maps, olfactory mosaic maps) with
those used for homing from unfamiliar sites (bicoordinate
navigation, gradient maps). However, attempts to develop a
unified theory have generally failed to address the differences in
the processing of sensory information required for these two types
of response. For piloting or place navigation, spatial
position in derived with respect to a configuration of familiar
landmarks or reference points, with the critical values being the
directions and, in some cases, distances to those reference points
(Figure 1A), or related features such as the size and appearance of
a visual landmark. A variant of this type of map, referred to
as a ōbearing mapö (Jacobs & Schenk,
2003), defines locations in terms of their compass bearing
(and distance?) from familiar reference points (Figure 1B). In contrast,
for navigation using a gradient map, spatial position is derived
from the difference between the local value of each map component
and the home value of that component (Figure 1C and D).
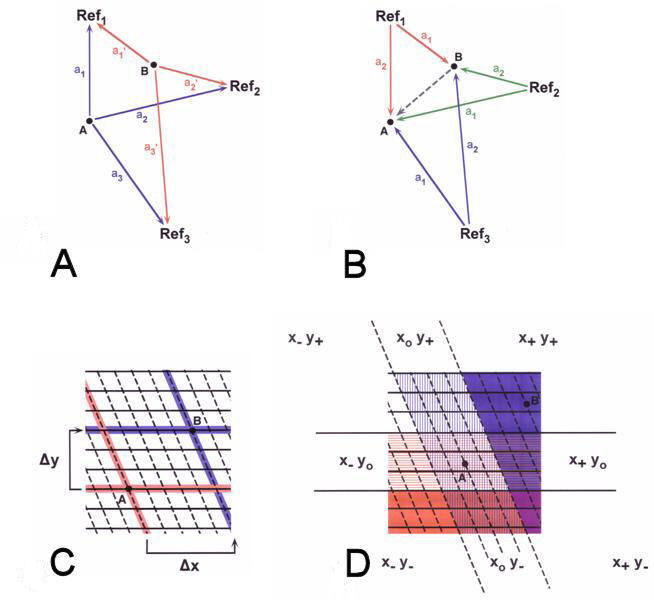 |
Figure 1. Maps of spatial
position. A) Place map, B) Bearing map, C) Bicoordinate
gradient map, D) Simple gradient map. æanÆ
indicates a measure of relative spatial relationship to the
organism at a given site (in A) or a compass bearing
from a familiar reference site (in B). ōxö and
ōyö are the values of two non-orthogonal environmental
gradients.
|
Because large scale
gradients of magnetic and olfactory cues are too weak for an animal
to detect directly, the alignment (and possibly steepness) of such
gradients have to be learned by obtaining multiple ōpoint
samplesö at different sites that are in a known spatial relationship
to one another (see Phillips & Deutschlander, 1987; Phillips, 1996). For
such large scale gradients of magnetic (Phillips, 1996) or olfactory
(Wallraff, 2004) cues, the origin or source of the gradient, and
thus the direction and distance from that source, is undefined in
the two dimensional world of the navigating animal.
Some types of cues may function in
both local place maps and large-scale gradient maps. For
example, local odor sources could be used in a ōmosaicö olfactory
map (comparable to a place map; Wallraff, 2004, see
also Bingman et al., this volume). However, this type of map is
limited in spatial scale by the detection range of discrete odor
sources, and the spatial heterogeneity of odor distributions (Ganzhorn, 1995; Waldvogel, 1987). Over a larger spatial scale,
other types of olfactory information (e.g., the ratio of odors from
extended sources; Wallraff, 2004) have been proposed to function in a
gradient map that can be extrapolated beyond an individualÆs area of
familiarity.
Spatial
variation in the magnetic field has also been suggested to provide map
information over different spatial scales (Phillips, 1996; Walker et al., 1997). Over
distances > 50-75 km (depending on the locality), regional
gradients in magnetic field components such as inclination, total
intensity and, possibly, declination could be used to provide unicoordinate or bicoordinate map information. At the other
extreme, local gradients extending over distances of < 5-10 km, which
often differ from the regional gradient in both direction and
steepness, may provide the basis for a small scale gradient map.
Recognizable local ōanomaliesö in the magnetic field may also provide
reference points (possibly in combination with visual or olfactory
cues) for a place or bearing map (Ganzhorn, 1990; Walker, Dennis, & Kirschvink,
2002). At
intermediate distances (from 10-20 km to 50-70 km), local and
regional variation tend to be similar in magnitude, so both higher
and lower magnetic field values are likely to be encountered in all
directions. At these distances, the magnetic field is
unlikely to provide a reliable source of map information (Phillips,
1996). Given the spatial heterogeneity of potential map cues,
animals that differ in mode of locomotion and range of movement are
likely to use different types of maps to determine spatial position.
Moreover, the same species may rely on different sources of map
information for short and long distance movements.
Interestingly, Schmidt-Koenig (1966; 1970) found that homing pigeons
at a loft in the United States and one in Germany showed markedly
poorer homing orientation from sites at intermediate distances from
the loft (30-70 km) in comparison to sites both closer and further
away (Figure 2), suggesting that different (and, at these sites,
non-overlapping) navigational mechanisms may be used for short and
long-distance homing (see also Matthews, 1963).
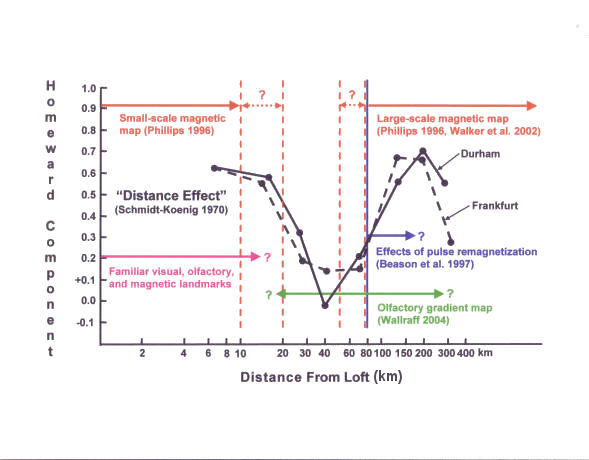 |
Figure 2.
Effects of distance on the accuracy of pigeon homing (data
from Schmidt-Koenig, 1970). Solid black symbols show
homeward component calculated from the pooled vanishing
bearings of birds released at four sites around a loft in
Durham, NC USA (solid black line) and a loft in Frankfurt,
Germany (dashed black line). Horizontal arrows show
predicted range of magnetic gradient maps (red), olfactory
gradient maps (green), familiar landmarks (purple), and
range of distances at which pulse remagnetization was found
to affect pigeon homing (blue). Vertical lines show
boundaries of predicted ranges of magnetic gradient maps
(red) and of range at which effects of pulse remagnetization
were observed (blue). Question marks indicate where
boundaries are not specified (green), dependent on
experience (purple), or data are not available (blue).
|
Gradient maps may also
differ in complexity (and, consequently, in underlying
neural architecture) in different animals. At one
extreme, a bicoordinate map may provide estimates of
actual position that vary continuously in both direction
and distance from the animalÆs ōhomeö or final
destination (bicoordinate position fixing; Figure 1C).
At the other extreme, a relatively simple
stimulus/response system that triggers a limited number
of discrete behaviors (ōorient to the southwest if the
values of both map coordinates are greater than the home
values"; i.e., X+Y+ in Figure 1D)
may suffice to produce accurate homing under some
conditions (K. Lohmann, personal communication).
A simple gradient map like that shown in
Figure 1D may be functionally indistinguishable from a
course-grained bearing map in which compass bearings are
associated with large areas or sectors around an
individualÆs home. Finally, sites that an animal
initially localizes by means of one type of map (e.g., a
magnetic or olfactory gradient map; Figure 1C) may
subsequently be incorporated into a grid of familiar
reference points (i.e., place map; Figure 1A), and/or
assigned a particular directional relationship to home
or other final destination (bearing map; dashed arrow in
Figure 1B; see Jacobs & Schenk, 2003).
Subsequent recognition of such sites may involve a
unique ōsignatureö of cues from the same, or a
different, sensory modality, including local olfactory,
visual, or magnetic cues (Burt, Holland, & Guilford,
1997; Ganzhorn, 1990; Walker et al., 2002). Like home/non-home
recognition cues, the role of cues in providing unique
labels or signatures of familiar sites (ōsite labeling"
cues) may be difficult to distinguish from that of cues
used to initially establish the spatial position of that
site (ōmap cuesö).
Both olfactory and magnetic cues have been proposed as
potential sources of gradient map information (Guilford et al., 1998; Hays
et al., 2003).
Olfactory effects on homing have been relatively easy to document,
although the question of whether olfactory cues are involved in the
map is more controversial. Magnetic field effects on
homing have been more difficult to demonstrate reliably. However,
there is a growing body of evidence for the magnetic fieldÆs
involvement in the map component of homing, due largely to the
development of experimental systems that have made it possible to
study map-based homing (true navigation) under controlled laboratory
conditions (Boles & Lohmann, 2002; Fischer, Freake, Borland, &
Phillips, 2001; Fischer, Munro, & Phillips, 2003; Lohmann, Lohmann,
Ehrhart, Bagley, & Swing, 2004; Phillips, Adler, & Borland, 1995;
Phillips, Freake, Fischer, & Borland, 2002b).
More generally,
attempts to understand the sensory basis of navigational gradient map(s) have been hampered by an overly simplistic view of animal
navigation which has largely ignored components of navigation systems
other than the map and compass. As a consequence, investigators
often fail to consider alternative hypotheses that involve, e.g.,
effects on cues used for home vs. non-home recognition, and for
recognition or ōlabelingö of familiar sites. By discussing such
alternatives, we hope to stimulate future studies that will clarify
the complex interactions of different types of sensory information
that underlie animal navigation and, specifically, the navigational
map or maps.
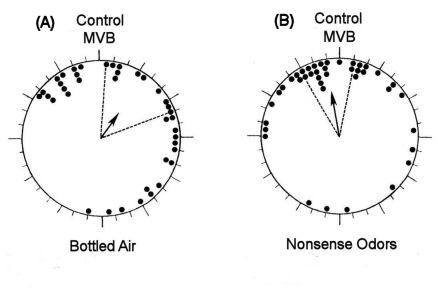 |
Figure 3. Effects of
exposure to bottled air and ōnonsense odorsö during displacement
to unfamiliar release sites on the homeward orientation of
pigeons. Vanishing bearings from four release sites
plotted as deviations from the mean vector bearing of controls
at each site. MVB=mean vanishing bearing (J.B. Phillips, J. Ganzhorn & K Schmidt-Koenig,
unpublished data)
|
A variety of
vertebrates have been shown to recognize and orient towards odors
associated with discrete food sources, nesting sites, or breeding
areas over distances from a few hundred meters to a kilometer or
more (e.g., Clark & Shah, 1992; Joly & Miaud, 1989,
1993; Nevitt, Reid, & Trathan, 2004).
In birds, discrete odor sources have also been hypothesized to play a
role in providing map information over longer distances, forming a
ōmosaicö map of olfactory space. In a mosaic olfactory map,
discrete odor sources function in the same manner as the reference
sites (landmarks) in a place map. Like distant visual landmarks,
it has been hypothesized that the location of discrete odor sources can
be determined without the individual actually visiting the site where
the odors originate by associating particular odors or combinations of
odors with winds blowing from different directions (but see
below). Even in birds, however, the range over which a mosaic
olfactory map can operate is likely to be limited to at most a few tens
of kilometers (Bingman et al., this volume; Ganzhorn, 1995; Waldvogel, 1987). For animals confined to the substrate, olfactory cues
are likely to function over considerably shorter distances, perhaps
only a few hundred meters (Joly & Miaud, 1989, 1993). This is
especially true in forested habitats where turbulence in the understory
eliminates any consistent relationship between overhead wind direction
and the direction of air currents at ground level (e.g., Baldocchi, 1989;
Hutchison & Hicks, 1985).
At the other extreme, Wallraff
(2004) proposed that birds may use an olfactory gradient map based
on the spatially varying ratio of certain atmospheric chemicals that
can be extrapolated to unfamiliar areas over distances of hundreds or
even thousands of kilometers.
A variety of
techniques have been used to investigate whether olfactory cues are
involved in the map component of navigation using pigeons as a model
organism, including: (1)
olfactory deprivation (plugging the nostrils, anesthesia or chemical ablation of the
olfactory mucosa, sectioning of the olfactory nerve, exposure to
filtered or bottled air during displacement to a release site, and
masking of natural odors with strong odorants), and (2) altering the
relationship between wind direction and natural or artificial odors
to which birds are exposed in their home loft, in some cases with
exposure to the same or different odors at an unfamiliar release
site prior to release.
Olfactory
deprivation either during displacement to a release site and/or
during the homeward journey has been shown to cause increased
scatter in initial homing orientation and/or decreased homing
success in a large number of experiments (e.g., Grant, Anderson, &
Twitty, 1968; Sinsch,
1990a, 1990b; Wallraff, 1990,
2004; Wallraff, Kiepenheuer, Neumann, & Streng, 1995). However, these effects are not
universal in pigeons or other animals (e.g., Ganzhorn, 1990, 1992; Sinsch, 1990b;
Wiltschko, Sch÷ps, & Kowalski, 1989). For example, in homing pigeons the effects of olfactory
deprivation have been shown to vary with loft location and/or early
experience, and with characteristics of individual release sites (Ganzhorn,
1990, 1992; Wiltschko et al., 1989). More importantly, experiments
showing effects of olfactory deprivation on homing have often failed
to rule out alternative explanations involving effects on sensory
systems other than olfaction (Mora, Davidson, Wild, & Walker, 2004), and/or olfactory
effects on component(s) of the birdÆs navigational system other than
the map (e.g., Ganzhorn, 1990; Phillip & Waldvogel, 1988; Wiltschko, 1996; and see below).
Two
alternative explanations for effects of olfactory deprivation on
homing warrant particular attention. In birds, a specialized magnetoreception system associated with the trigeminal nerve has been
implicated in the map component of homing (e.g., Beason & Semm, 1987,
1996; Beason, Wiltschko, & Wiltschko, 1997; Munro et al., 1997a; Munro, Munro,
Phillips, & Wiltschko, 1997b; Walker et al.,
1997; and see below). In conditioning experiments with pigeons,
Mora et al. (2004) found that anesthesia of the olfactory mucosa, a
technique used to produce olfactory deprivation in behavioral studies
of pigeon homing (see Wallraff, 2004), also blocks trigeminal nerve
mediated responses to magnetic stimuli. Consequently, studies of
homing orientation using this technique may inadvertently deprive birds
of magnetic, as well as olfactory, information. Secondly,
deprivation of olfactory information may deprive birds of the ability
to distinguish home from non-home, or to recognize the olfactory
signature of familiar sites (see earlier discussion). The
possibility of such effects is suggested by preliminary experiments
with homing pigeons (Phillips, Ganzhorn, & Schmidt-Koenig,
unpublished data). Previous experiments have shown that
preventing access to olfactory cues during displacement to an
unfamiliar release site can, under some conditions, affect the homing
orientation of pigeons (e.g., Papi, 1990;
Wallraff, 2004; Wiltschko et al., 1989). In an attempt to distinguish map-related and
non-map-related effects of olfactory deprivation, experienced homing
pigeons were subjected to one of three treatments during displacement
to four release sites arrayed symmetrically around the home loft.
Controls were exposed to natural odors during displacement. One
experimental group was exposed to synthetic bottled air during
transport that eliminated access to natural odors (ōbottled airö
group). A second experimental group was also exposed to synthetic
bottled air during displacement, but a series of artificial odors
(peppermint, spearmint, orange, clove, etc.) was introduced into the
air stream (ōnonsense odorö group). A new odor was introduced every five
minutes during the displacement to prevent habituation. The same
sequence of artificial odors was presented to the birds during
transport to each of the release sites, so that even if one of the
nonsense odors occurred naturally in a particular direction from the
home loft, exposure to this odor would not produce consistent homeward
orientation when departure directions were pooled from all four release
sites.
Figure
3A & B shows the vanishing bearings (ōinitial orientationö) of the
two experimental treatments plotted as deviations from the mean
vector bearing of controls tested at the same release site. Plotting the
bearings in this way eliminates any effect of site-specific
deviations from the home direction. The distribution of vanishing
bearing from bottled air birds differed significantly from that of
controls (Figure 3A). In contrast, the distribution of
vanishing bearings from nonsense odor
birds was indistinguishable from that of controls (Figure 3B). While by
no means conclusive [Footnote 1],
these findings illustrate the importance of carrying out experiments
to determine whether exposure to unfamiliar odors that provide no
information about the position of the release site relative to the
home loft mimics the effect of exposure to natural odors [Footnote 2].
If so, this would suggest that olfactory cues play a role in a
component(s) of the animalÆs navigation system other than the map.
For example, exposure to non-home odors could activate the birdsÆ
navigational system, causing the birds to access stored map and
compass information and/or to acquire non-olfactory
information necessary for homing. The crucial point here is that
despite the large number of experiments showing effects of olfactory
deprivation on homing (for reviews see Papi, 1990; Wallraff, 2004),
critical controls have not been carried out to determine whether
these effects are due to the involvement of olfactory cues in the
map [Footnote 3].
The same criticism can be leveled at experiments comparing the
orientation of groups of pigeons exposed to natural odors at a
ōfalseö release site and then rendered anosmic before being transported to a site in the opposite
direction from the home loft for testing (e.g., Benvenuti & Wallraff,
1985; Kiepenheuer, 1985).
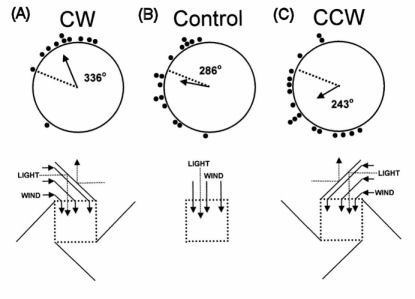 |
Figure 4. Initial orientation of homing
pigeons after housing in deflector and control lofts (data
from Waldvogel & Phillips, 1982). A) Initial orientation of
pigeons housed in deflector loft that produced a clockwise (CW)
rotation of wind direction and reflected light cues (see
inset). B) Initial orientation of pigeons housed in control
loft without deflector panels. C) Initial orientation of
pigeons housed in deflector loft that produced a
counterclockwise (CCW) rotation of wind direction and
reflected light cues (inset). |
In addition to
olfactory deprivation, a number of experiments with homing pigeons
using techniques that alter the relationship between wind direction
and either natural or artificial odors have produced findings
consistent with the olfactory map hypothesis. However, like the olfactory
deprivation experiments, most, if not all, of these
experimental findings are open to alternative interpretations. Here
we focus on one type of such experiments, the so-called
ōdeflector loftö experiments. In these experiments, three lofts
were constructed with sides of wire mesh and vertical louvers that
allowed air to pass through unimpeded. Two of the lofts were
equipped with large ōdeflector panelsö attached to the sides that
rotated incoming winds in either a clockwise (CW) or
counterclockwise (CCW) direction (Figure 4). We focus on the
deflector loft experiments for several reasons: (1) birds are exposed to
natural, rather than artificial, odors, (2) shifts in wind direction
are produced passively, thus avoiding potential artifacts associated
with fans (noise, infrasound, turbulence, electromagnetic fields, etc), (3) the
predicted effects of the deflector lofts are unambiguous, i.e., if
pigeons learn the distribution of odor sources by associating the
presence of specific odors with winds arriving from particular
directions, then rotation of wind direction in the deflector lofts
must produce a corresponding shift in the birdsÆ olfactory
map and, therefore, in the direction of homing orientation (Phillips
& Waldvogel, 1982), and (4) the deflector loft effect has been
replicated by laboratories in Italy, Germany and the
United States (e.g., Baldaccini, Benvenuti, Fiaschi, & Papi, 1975; IoalĶ, Papi, Fiaschi,
& Baldaccini, 1978; Kiepenheuer,
1978; Waldvogel, Benvenuti, Keeton, & Papi, 1978).
Although the
deflector loft experiments are frequently cited as support for the
olfactory map hypothesis (Wallraff, 2004), several findings call this
conclusion into question.
(1) Kiepenheuer (1979)
prevented deflector loft birds
from detecting olfactory cues by anesthetizing
the olfactory mucosa or plugging the
nostrils prior to displacement to an unfamiliar release site. Despite their inability to detect natural odors, the
birds exhibited shifted orientation indistinguishable from that of
pigeons that had been housed in the deflector lofts but were able to detect
natural odors. These findings indicate that the deflection of
initial orientation of deflector loft birds is not a response to
olfactory cues.
(2) Kiepenheuer (1982) removed the vertical
louvers from the sides of the deflector lofts (leaving only wire
mesh and the deflector panels), resulting in a ōwhirlwindö pattern
of wind flow within the lofts that eliminated any consistent
relationship between prevailing wind direction and the wind
direction experienced by birds at different locations within
the loft. Again the deflection of initial orientation was
unaffected, suggesting that associating odors with wind direction does not play a role in the deflector loft
effect.
(3) Phillips & Waldvogel (1982)
showed that the deflected initial orientation of pigeons held in
deflector lofts for five to seven days (ōshort-termö residents) was a response
to light cues reflected from the deflector panels, rather than
wind-born cues. The deflected orientation of ōpermanent residentö
birds was also shown to be a response to the altered pattern of light cues
visible to the birds in the deflector lofts, rather than
wind-borne odors (Phillips & Waldvogel, 1991). Phillips & Waldvogel
(1982, 1988) presented evidence that the deflector loft effect
results from rotation of a polarized-light-based reference system
used to calibrate the sun compass and, possibly also, the magnetic
compass of pigeons. Subsequent experiments with
migratory birds have demonstrated the use of polarized light cues to
calibrate the sun compass (Phillips & Moore, 1992) and magnetic
compass (Muheim, Moore, & Phillips, 2006a; Muheim, et al., 2006b).
(4) Waldvogel
and Phillips (1982) found that the shift in the initial orientation of
permanent resident deflector loft birds was present when birds were
released under sunny skies, but not under overcast skies. Since pigeons
are unlikely to rely on different source(s) of map information under
sunny and overcast conditions, these findings provide further evidence
that the deflector loft effect is linked to the use of the sun compass,
rather than an olfactory map.
The results of
these four experiments demonstrate: a) that the deflector loft
effect is caused by rotation of light, rather than olfactory, cues, and
b) that wind-borne cues perceived in the home loft do not play a
role in the navigational system of pigeons, at least under the
conditions used in these experiments. Despite the consistency of
the findings outlined above, the results of a fifth type of
deflector loft experiment has been widely cited as evidence for an
olfactory map.
In pigeons,
olfactory input projects to the ipsalateral hemisphere and from
there, via
the anterior commissure, to the contralateral hemisphere. Foa,
Bagnoli, and Giongo (1986) sectioned the anterior commissure of homing pigeons,
restricting olfactory input from each nostril to one hemisphere.
After recovery, ōsplit brainö birds were housed alternately in the
CW and CCW deflector lofts. While housed in the CW loft, the
birdsÆ right nostrils were plugged, so olfactory information reached
only the left hemispheres. While housed in the CCW loft, the birdsÆ
left nostrils were plugged, so olfactory information reached only
the right hemispheres. When the birds were released at an
unfamiliar site with their right nostrils plugged, their initial
orientation was deflected CW. When the birds were
released with their left nostrils plugged, their initial orientation
was deflected CCW. These findings are consistent with
the birds having established independent CW- and CCW- rotated
olfactory maps on the two sides of the brain (FoÓ et al., 1986), and
appear to contradict those of the four experiments described above.
However, like the effects of olfactory
deprivation, there are a number of alternative explanations
for the results of the ōsplit brainö experiments. Here we discuss
one such possibility. As discussed earlier, olfactory cues may play a role in
a component of the pigeonÆs navigational system other than the map,
such as distinguishing home from non-home (Figure 3). We propose that
calibration of the pigeonÆs sun compass (which is altered by
light cues in the deflector lofts; see earlier discussion), and
access to this stored calibration information, is triggered by
exposure to home and non-home odors (respectively). According to
this scenario, the effects of plugging the right or left nostril in
the split brain experiments are a consequence of storing and
accessing differently calibrated sun compass systems on the two
sides of the brain. In contrast to an effect on the olfactory map
(as
proposed by FoÓ et al., 1986), this explanation reconciles the split
brain experiments with the earlier deflector loft results.
Evidence that the deflector loft
effect is not due to wind-borne odors appears to be contradicted by
other ōwind redirectionö experiments with homing pigeons (reviewed
by Papi, 1990; Wallraff, 2004). However, here again, alternative
hypotheses involving both olfactory and non-olfactory cues have been
largely ignored. For example, the effects observed in these
experiments could be caused by: a) light cues
reflected from glass or Plexiglas panels (Phillips & Waldvogel,
1988), b) aversive stimuli associated with electric fans
(electromagnetic fields, air turbulence, infrasound, etc.), or c)
pigeons using distinctive odors to ōlabelö wind direction [Footnote 4].
In summary, the
literature does not provide compelling evidence for the involvement
of olfactory cues in a long-distance gradient map. In birds like
homing pigeons, there remains the possibility that olfactory cues
play a role in a short-distance ōmosaicö map, comparable to a place
or bearing map (Ganzhorn, 1990, 1992). However, the findings of the
deflector loft experiments make it unlikely that either type of
olfactory map involves natural wind borne odors reaching pigeons in their
home loft. As in the examples discussed
earlier, other experiments that have provided evidence consistent with an
olfactory mosaic map require additional controls to rule out
alternative explanations involving effects on both olfactory and
non-olfactory cues. Particular attention should be paid to the
possibility that specific odors encountered during displacement to,
or upon arrival at, a release site may provide one type of ōsite
labelingö cues that help identify previously visited localities or
regions (Ganzhorn, 1990) and/or distinguish home from non-home (Figure 3), rather that providing remote (i.e.,
wind-borne) information about the location of distant odor sources
(see earlier discussion). Clearly, a better understanding of the
role of olfactory cues in both map and non-map components of animal
navigation systems is crucial to interpreting the effects of a
variety of experimental treatments, including ablations of brain
regions involved in processing both spatial and olfactory
information (e.g., Gagliardo, Ioalķ, & Bingman, 1999; Gagliardo,
Ioalķ, Odetti, Kahn, & Bingman, 2004; Gagliardo, Mazzotto, & Bingman,
1997; Bingman, Gagliardo, & IoalĶ, 1996; Ioalķ, Gagliardo, & Bingman,
2000).
On a global scale, magnetic
field intensity currently varies from a minimum of 20,000-25,000 nT at the magnetic equator to a maximum of
60,000-65,000 nT at the poles, and magnetic inclination varies from
0o (horizontal) at the magnetic equator to 90o
(vertical) at the poles. Yeagley (1947, 1951) was the first
to investigate the possibility that spatial variation in the magnetic field could
provide homing pigeons with a source of north-south position (see also
Gould, 1985; Moore, 1980; Walcott, 1991). However, there are a number of
potential problems with the use of a magnetic map, especially for
organisms that must resolve differences in spatial position on the
order of a kilometer or less (Phillips, 1996).
(1) Magnetic field gradients are
extremely weak, i.e., variation in total intensity averages only
~5-10 nT/km (~0.01% of the total field intensity) and in inclination only ~0.010 o /km. The
weakness of the magnetic gradients has several important
implications for the design of animal navigation systems: a) the
sensory mechanism(s) responsible for detection of spatial variation
in the magnetic field must be extraordinarily sensitive (Phillips,
1996), b) magnetic gradients can not be detected directly, but
instead must be derived from a series of ōpoint samplesö [i.e.,
isolated measurements that contain no information about the
alignment of the gradient(s) at the measurement site but are in a
known spatial relationship to one another (Phillips & Deutschlander,
1997)], c) the organism must have an independent means of estimating
geographic position within its area of familiarity (e.g., a place
map, or bearing map), or a non-map-based system for determining its
spatial position (e.g., a path integration system; Wiltschko &
Wiltschko, 2000, 2003), in order to determine the spatial
relationship among measurement sites, d) the organism must be able
to store precise measurements of magnetic field component(s) in
memory to compare the values obtained at different sites within the
spatial array, and e) because an organism must sample multiple sites to
build up a knowledge of spatial variation in the magnetic field, use
of a gradient magnetic map will be dependent on experience.
That is, there
should be an ontogenetic progression from reliance on familiar
configurations of landmarks (a place or bearing map) and/or path
integration, to reliance on a full blown gradient map (Wiltschko &
Wiltschko, 2000, 2003).
(2) Due to difference in the iron
content of the underlying rock layers, local gradients in the
magnetic field often differ in direction and/or steepness from the
regional gradient. These local irregularities in the magnetic field
will introduce large errors in map estimates if extrapolation of
the local gradients is used to estimate geographic position after
long distance displacement. As discussed earlier, a gradient map may
be used at some (but not all) sites for short distance movements
within the range of the local gradients (< 5-10 km). A gradient
magnetic map may also be used for movements over long distances
(>50-75 km) where the regional gradient predominates. For such a
large-scale map, the spatial variation that occurs at distances of
less than ~ 50-75 km (i.e., variation due to local gradients) would
constitute noise. Consequently, such a large-scale magnetic map
could only be used to locate an area 50-75 km in diameter,
rather than a specific site within this area (Phillips, 1996;
Walker et al., 2002). At intermediate distances from 10-20 km to 50-75 km,
where the contribution of local and regional spatial variation is
similar in magnitude, there is unlikely to be a spatial
signal that could used to provide a meaningful estimate of
geographic position. Therefore, the geomagnetic field is likely to
be used either for a short-distance, high-resolution map or for a
long-distance, low-resolution map, but not for a map that functions at
intermediate distances (Figure 2). Consequently, animals that navigate over
long distances may be forced to rely on different sources of map
information
and/or different types of spatial information (place or bearing vs.
gradient maps) for different scales of movement.
3) Regular temporal variation in
the magnetic field that tends to be greatest during the day light hours could introduce
significant errors in fine scale map estimates. For animals that
use the magnetic field for a short distance (high resolution) map,
therefore, strategies such as averaging multiple measurements over
extended periods of time and/or taking measurements at night when
temporal variation in the magnetic field is reduced may be necessary
to minimize this source of error (Phillips, 1996; Rodda, 1984).
(4) Magnetic storms produce large
and unpredictable fluctuations in the magnetic field that could
introduce large errors in estimates of map position, especially in
animals using a short-distance, high resolution map. One way
to minimize such errors would be to use characteristics of magnetic storm activity, e.g., rapid
fluctuations in the geomagnetic field that occur during magnetic
storms, to avoid taking map measurements when the field is unstable
(Phillips, 1996; Phillips & Deutschlander, 1997).
(5) Finally, long term variation in
the magnetic field could introduce systematic errors when
significant
periods of time separate displacement into unfamiliar territory and
the return back to the origin of that displacement, e.g., intervals
of several years that occur in some species between juvenile
dispersal from a natal site, and the return migration of adults to
the natal site to breed (Courtillot et al., 1997). Strategies such
as taking measurements immediately before and after displacements
(or during the nights immediately before and after displacement) to
minimize the effects of temporal variation, and/or factoring out the
regular component of such variation based on measurements obtained
during intervals between bouts of movement would be necessary to
prevent map coordinates from ōdriftingö over time due to such long
term changes.
Despite these
very real difficulties, there is a growing body of evidence for the
use of a magnetic map for both short-distance and long-distance
homing. ōRoute-deprivationö studies [Footnote 5]
of homing orientation by newts (Phillips et al.,
1995), juvenile alligators (Rodda, 1984), sea turtles (Lohmann et
al., 2004), homing pigeons (Walcott & Schmidt-Koenig, 1973) and
spiny lobsters (Boles
& Lohmann, 2003) show that these animals are able to
use map information obtained at the testing site to determine
geographic ("map") position. Newts provide an especially interesting case.
As discussed previously, in woodland habitats where newts occur,
turbulence in the understory eliminates any consistent relationship
between prevailing wind direction and the direction of air currents
at ground level (Baldocchi, 1989; Hutschison &
Hicks, 1985), thus eliminating any possibility
that newts use wind-borne odors to learn the distribution of distant
odor sources. The magnetic field, therefore, appears to be the only
potential source of gradient map information available to newts, and
other similar organisms, that live in this type of habitat.
Nevertheless, showing magnetic field effects on homing orientation
is not sufficient to establish the use of a magnetic map. Magnetic
compass (as opposed to map) cues have been shown to play a role in
two important aspects of homing. (1) Young inexperienced
individuals may rely on the magnetic compass to determine
displacement direction (i.e., path integration; Wiltschko &
Wiltschko, 2000, 2003) before they acquire sufficient experience to
learn the distribution of map cues. (2) Magnetic compass
information may be used for the compass component of homing, e.g.,
when cloud cover restricts access to celestial compass cues (Diego-Rasilla,
Luengo, & Phillips, 2005; IoalĶ, 1984).
Two lines
of evidence indicate that, in addition to providing compass
information, the magnetic field is an important source of map
information: (1) amphibians and birds have two distinct
magnetoreception mechanisms, one that provides compass information
and one that plays a specialized role in the map component
of homing, and (2) under some conditions, experienced adult
amphibians, reptiles and birds respond to small magnetic field
changes as if they have been displaced to a new location (ōsimulated
magnetic displacementsö).
Magnetic compass orientation in amphibians and birds has been shown
to be sensitive to the presence and wavelength of light (Deutschlander,
Borland, & Phillips, 1999; Muheim, Bõckman, & ┼kesson, 2002; Phillips
& Borland, 1992a, 1992b; Wiltschko, Munro, Ford, & Wiltschko,
1993; Wiltschko & Wiltschko, 1995b, 1998, 1999, 2001),
consistent with theoretical models of the mechanism
of magnetoreception that implicates a photoexcited radical pair
reaction occurring in a specialized photoreceptor (Ritz, Adem, &
Schulten, 2000). Both frogs and salamanders trained to exhibit shoreward
orientation under natural light oriented in the trained direction
when tested under full spectrum light,
but exhibited 90o
shifted orientation under wavelengths of light > 500 nm
(Freake & Phillips, 2005; Phillips & Borland, 1992a, 1992b). Control
experiments in which newts were trained under wavelengths >
500 nm and tested under either full spectrum or long-wavelength
light indicate that the 90o shift results from a
direction effect of light on the underlying magnetoreception
mechanism (Phillips & Borland, 1992a, 1992b). In newts, the 90o shift is the result of an
antagonistic interaction between short-wavelength (< 450 nm)
and long-wavelength (> 500 nm) inputs mediated by extraocular
photoreceptors located in or near the pineal organ (Deutschlander et
al., 1999; Phillips &
Borland, 1992a, 1992b). Magnetic compass
orientation in birds shows a complex dependence on the wavelength
and intensity of light (Wiltschko & Wiltschko, 2001). Unlike
amphibians, the light-dependent magnetic compass is mediated by
photoreceptors in the retina. As found in a number of visual
mechanisms in birds (e.g., Clayton & Krebbs, 1994), the
light-dependent magnetic compass is strongly lateralized, involving
only the right retina (Wiltschko, Traudt, G³nt³rk³n, Prior, &
Wiltschko, 2002b; Wiltschko, Munro, Ford, & Wiltschko, 2003). Recent experiments showing effects of low-level radio
frequency radiation on magnetic compass orientation in birds (Ritz,
Thalau, Phillips, Wiltschko, & Wiltschko, 2004) are consistent with the energy state transitions
predicted to occur in a mechanism involving a photo-excited radical
pair reaction (Ritz et al., 2000).
In
addition to the light-dependent magnetic compass, both amphibians and
birds have a second, non-light-dependent mechanism utilizing a
permanent magnetic material that is most likely biogenic magnetite
(Munro et al., 1997a, 1997b; Phillips, Borland, Freake, Brassart, & Kirschvink,
2002a; Wiltschko, Munro, Wiltschko,
& Kirschvink, 2002a). In birds, the magnetite-based receptor
appears to be associated with the trigeminal nerve system (Beason &
Semm, 1987; Fleissner et al., 2003). A similar system has been
characterized in salmonid fish, although its role in behavior remains
to be established (Walker et al., 1997). The magnetite-based
receptor in birds and amphibians is not involved in magnetic compass
orientation, but instead is brought ōon lineö when these animals
navigate using map-based cues (Beason & Semm, 1996; Beason et al., 1997;
Munro et al., 1997a, 1997b; Phillips, 1986a; Phillips & Borland,
1994; Phillips et al., 2002a). In birds, evidence for a
magnetite-based receptorÆs involvement in the navigational map has come
from pulse remagnetization experiments, in
which animals are exposed to a rapid,
high intensity magnetic pulse that is strong enough to remagnetize
single domain magnetite particles, but with no lasting effects on a
photoreceptor-based mechanism involving a radical pair mechanism.
Munro
et al.(1997a,
1997b)
found that pulse remagnetization altered the migratory orientation
of experienced adult birds that navigate using both map and compass
information, but not that of na’ve, young birds that undertake their
first migration using only compass information (Perdeck, 1958).
Similarly, pulse remagnetization was found to affect the initial
orientation of homing pigeons, but only at sites > 80 km from
the home loft (Beason et al., 1997; see Figure 2). The absence of an effect on
pigeons released at shorter distances is consistent with the use of a large
scale, low resolution magnetic map.
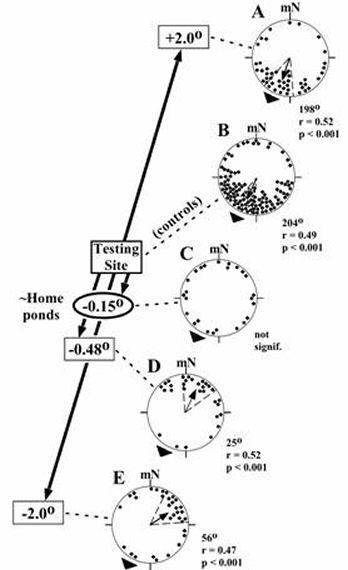 |
Figure 5. Effects of small changes in magnetic
inclination on homing orientation of Eastern red-spotted newts
(data from Fischer et al., 2001; Phillips et al., 2002b).
Magnetic bearings pooled from newts tested in one of four
horizontal alignments of the magnetic field (magnetic North =
north, east, west or south). Values at the left indicate
the changes in inclination to which the newts were exposed
relative to the ambient value at the testing site (controls).
Changes in inclination were made with little or no change in the
total intensity. Newts were collected from ponds located
~42 km south-southwest of the testing site (home direction = 207o).
Magnetic inclination at the ōhome pondsö was approximately 0.17o
less than the inclination at the testing site. mN =
magnetic north. |
|
Experiments with
newts (Fischer et al., 2001; Phillips et al., 2002b), sea turtles (Lohmann
et al., 2004) and migratory birds (Fischer et al., 2003) have been
carried out to investigate the magnetic fieldÆs role in providing
gradient map information. In these experiments, subjects were
exposed to different values of one or more magnetic field components
similar to those found at locations north and south of the testing
site (ōsimulated magnetic displacementsö). Both newts and sea
turtles exhibited opposite directions of orientation corresponding
to the relative positions of the simulated magnetic map
coordinates.
In initial experiments with
newts, the inclination of the magnetic field was changed by ▒ 2░
Figure 5a,e). In subsequent experiments, smaller changes in
the magnetic field inclination were used to ōtitrateö the home
value. Consistent with the magnetic map hypothesis, reversal
of the newtsÆ homing orientation bracketed the home value of
magnetic inclination (Figure 5b,d) and newts exposed to the home
value of inclination failed to show a consistent direction of
orientation (Figure 5c). To rule out the possibility that the
changes in magnetic inclination affected the magnetic compass, newts
exhibiting shoreward magnetic compass orientation, which does not
require map information, were exposed to ▒ 2░ changes in inclination
(Fischer et al., 2001). There was no effect on shoreward
magnetic compass orientation, indicating that the magnetic compass
was unaffected.
Simulated
magnetic displacement studies of Tasmanian silvereyes also point to
a possible role of the magnetic field in providing map information,
although the findings are less clear than those observed in newts
and sea turtles. During the fall migration, Fischer et al. (2003)
captured adult silvereyes en route from Tasmania northward
along the southeast coast of Australia. In initial baseline
tests, carried out in the ambient field of Armidale, NSW, silvereyes
oriented in a seasonally appropriate north-northeasterly direction.
The silvereyes were then divided into two groups and exposed to values of magnetic field intensity and
inclination that would normally be encountered to the south
(simulated south displacement; SimS) and to the north (simulated
north displacement; SimN) of the testing site. The northern values
correspond to locations either within (intensity), or to the north
of (inclination), the silvereyeÆs winter range. The orientation of
the two groups of birds was then tested on alternate days to obtain
five to six directional responses from each bird. Birds exposed to the
SimS
displacement continued to show northeasterly magnetic orientation
similar to their control responses, while birds exposed to the SimN
displacement failed to show a significant direction of orientation
and differed significantly from their control responses, as well as
from the responses of the birds exposed to the SimS condition
(Fisher et al., 2003).
Fischer et
al. (2003) discussed several possible explanations for the failure of
birds in the SimN condition to exhibit a consistent direction of
orientation. However, a recent reanalysis of these data suggests
that rather than a failure to orient (or a failure of individual
birds to agree on a single direction of orientation), birds in the
SimN condition may have exhibited a two stage response. Figure
6
shows the orientation of birds in the SimS and SimN conditions
during the first three days (Figure 6, left diagrams) and the second
2-3 days (Figure 6, right diagrams) of testing. There was no
difference in the orientation of birds in the SimS condition during
these two time periods (Figure 6, bottom diagrams; p>0.10, Hotelling's two-sample test). However, birds in
the SimN condition oriented to the east during the first three
nights (Figure 6, top left diagram; p<0.05, Hotelling's one-sample
test), and then exhibited a significant
shift in the direction of orientation (p<0.05, Hotelling's
two-sample test), resulting in a directional
tendency (p<0.10) opposite that of the birds in the SimS birds (Figure
6, bottom
right diagram).
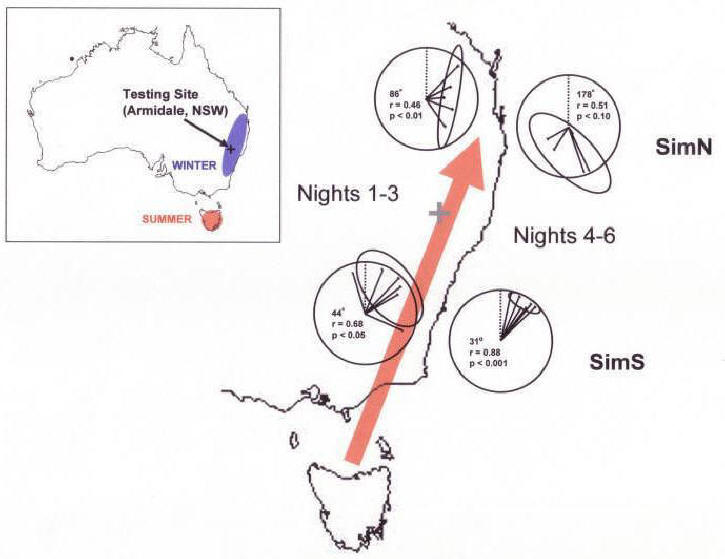 |
Figure 6. Migratory orientation of Tasmanian silvereyes
after exposure to simulated north (SimN) and south (SimS)
magnetic displacements. Black outline shows the coast of
Tasmania (bottom of figure) and of southeastern Australia.
Lower circular diagrams show the orientation of birds exposed to
magnetic field values normally encountered to the south near the
start of the Fall migration (simulated south displacement, or
SimS). Upper circular diagrams show the orientation of birds
exposed to magnetic field values normally encountered at the
northern edge of, or to the north of, the winter range
(simulated north displacement or SimN). Left circular diagrams
of each pair show data from nights 1-3 of testing. Right
circular diagrams show data from nights 4-5 (SimS) or 4-6 (SimN)
of testing. Lines originating from the center of each diagram
are mean vectors calculated for individual birds from its
nightly bearings. Ellipses are 95% confidence intervals for
each distribution. Statistics (mean vector bearing, mean vector
length, and probability level) are for the HotellingÆs test (Batschelet,
1981) used to test for departure from a random distribution.
Gray cross shows the approximate location of the testing site.
Inset: Map of Australia showing breeding range of this
population in Tasmania (red) and the approximate winter range
along the southeastern coast of Australia (blue). Testing site
(Armidale, NSW) is indicated by a black cross. |
|
Mouritsen (2003) proposed that
migratory birds use a unicoordinate map to determine
north-south position, and rely on major topographic features
(rather than a second map coordinate) to determine their east-west
position. In southeastern Australia, silvereyes migrate along a
narrow corridor parallel to the coastline that runs roughly north-northeast/ south-southwest. Consistent with MouritsenÆs
suggestion, the tendency of birds in the SimN condition to fly to
the east would have enabled them to locate the coastline and,
therefore, to determine their east-west position. The subsequent
tendency of the birds to orient in a southerly direction, i.e.,
opposite that of birds in the SimS condition, is consistent with the
birds interpreting their geographic position as being to the north
of their winter range. In other words, these findings suggest that
the birds may be able to derive their north-south
position from the geomagnetic field.
Consistent
with a map effect, more recent experiments using somewhat larger
magnetic field changes have found an affect of the SimN (but not SimS)
condition on the orientation of experienced adult birds, but not that
of na’ve young birds captured on the breeding grounds shortly after
fledging and prior to their first migration (Deutschlander et al., in
preparation). Birds migrating for the first time rely on a
ōvector strategyö that combines compass information with a temporal
program (Berthold, 1990, 1991; Perdeck, 1958), rather than
true navigation that requires both a map and compass. Other than the
use of map information, the physiological state of young inexperienced
and experienced adult migrants appears to be quite similar (increased
fat reserves, nocturnal restlessness, etc.). Consequently, these
findings suggest that, rather than an effect on the compass, the
response of experienced birds in the SimN condition is due to an effect
on the map.
Can the findings of the simulated
magnetic displacements be explained by an effect on a component of
animalsÆ navigation system other than the map? In these experiments, subjects were
exposed to a single magnetic field (equivalent to a ōpoint sampleö
discussed earlier) and, thus, were unlikely to interpret the altered
magnetic field as a familiar magnetic ōlandmarkö or reference point;
use of magnetic landmarks would require recognition of unique
spatially varying features of local magnetic topography (i.e.,
directions and rates of change in one or more magnetic field
components; Walker et al., 2002). Moreover, subjects in the simulated
magnetic displacement experiments were exposed to different magnetic
field conditions at the same site, so other potential map cues such
as odors were held unchanged. Consequently, the findings of these
experiments cannot be explained by assuming that the altered magnetic
fields caused the experimental subjects to attend to
non-magnetic (e.g., olfactory) map cues. The most likely
explanation for the findings of the magnetic displacement
experiments, therefore, is an effect on a magnetic gradient map.
A major unresolved issue is
whether short-distance migrants like newts, and/or long distance
migrants like sea turtles and many species of birds, have unicoordinate or bicoordinate maps. Simulated
magnetic displacement experiments with newts, sea turtles and migratory
birds have provided evidence for the use of the magnetic field to
derive a map coordinate that provides information about geographic
position along roughly the north-south axis. Regional gradients
in magnetic field components such as inclination and intensity run
more-or-less north-south and, thus, are well suited to provide
information about geographic position along this axis. It remains
to be determined whether these animals are able to determine their
geographic position along a second, non-parallel (east-west) axis.
If these animals are able to use a
second map coordinate, what might this coordinate be? In many parts
of the world, large-scale variation in magnetic declination (the
angle between magnetic and geographic north) varies along a gradient
that is roughly orthogonal to the gradients of intensity and
inclination and, thus, provides a possible second map coordinate.
Birds have been proposed to derive geographic North as a reference
for calibration of their compass systems by averaging the positions
of the band of maximum skylight polarization at sunrise and sunset
(Muheim, et al., 2006b; Phillips & Waldvogel, 1982, 1988). It remains to be determined
whether measurements of magnetic declination using such a reference
system would be accurate enough for a large-scale
map. Nevertheless, for long-distance migrants like silvereyes, a
second map coordinate derived from magnetic declination remains a
possibility. For example, silvereyes migrating from Tasmania
northward along the coast of eastern Australia travel in a corridor
approximately 200 km wide. Magnetic declination changes by about
two
degrees over this distance. Since the gradient of magnetic
declination is roughly perpendicular to the gradients of intensity
and inclination, silvereyes provide an excellent model system in
which to investigate the possibility of a bicoordinate magnetic
map. Use of a bicoordinate magnetic map is not ruled out by the
experiments described earlier in which silvereyes appeared use an
alternative strategy (ōfly east until you encounter the coastlineö)
to determine east-west position (Figure 6). The birds in these
experiments were housed and tested indoors without access to
celestial cues and, thus, could not have used a reference system
based on celestial polarized patterns to obtain measurements of
magnetic declination (see earlier discussion).
Bicoordinate
navigation is even more problematic for animals that require short
distance, high resolution map information (i.e., for movements over
distance <5-10 km). At such short distances, it is unlikely
that magnetic declination could to be used to derive a second map
coordinate, since the required accuracy would be roughly 100 times
greater than that required by, e.g., a migratory bird, and the forest
canopy would limit the access of newts and other inhabitants of the
forest floor to celestial cues such as polarized light patterns that
could be used to derive a geographic reference (Muheim, et al.,
2006b). An alternative possibility is that local
variation in the magnetic field results in non-parallel gradients of
components such as inclination and intensity that do not require an
independent directional reference. However, local gradients
suitable for bicoordinate navigation are likely to be present at some
sites, but not at others (Phillips, 1996). If so, a bicoordinate
magnetic map cannot provide a universal solution to the problem of
navigation over small spatial scales. An interesting question is
whether newts, and other organisms that require short-range, high
resolution map information, are found at higher population
densities in areas where local magnetic field gradients are suitable
for bicoordinate navigation (S.C. Borland, personal communication).
We have argued for a fundamental
distinction between local ōplaceö maps (and related maps that
involve familiar reference sites, including bearing maps), and
larger scale gradient maps that can be extrapolated beyond an
individualÆs area of familiarity. Not only are these two categories
of maps likely to involve different types of sensory information,
but also different processing mechanisms and, consequently,
different populations of neurons in the central nervous system.
Contrary to a number of recent authors (Bingman et al., this volume; Jacobs & Schenk, 2003; Wallraff,
2004), we also conclude that
there is no compelling evidence for the involvement of olfactory
cues in a large-scale gradient map. In contrast, a compelling case
is emerging for the use of magnetic cues to derive at least one map
coordinate. Both amphibians and birds have a magnetoreception
mechanism distinct from the magnetic compass that appears to play a
specialized role in the map component of homing. Simulated magnetic
displacement experiments have provided evidence that amphibians,
reptiles and birds (see also recent findings from spiny lobsters;
Boles & Lohmann, 2003) will, at least under some conditions,
interpret small changes in the magnetic field as changes in
geographic position. Nevertheless, magnetic navigation does not
provide a universal solution to the problem of map-based homing,
i.e., the magnetic field does not provide useful map information at
all sites or at all spatial scales.
The
emerging picture from studies of animal navigation
systems is one of a patchwork of mechanisms that vary in
utility depending on local environment (e.g.,
the characteristics of local gradients), range of movement, and
individual age/experience. Map information involving multiple sensory
modalities may be involved, including both olfactory and magnetic
cues, as well as both map-based and route-based mechanisms for
deriving spatial position at unfamiliar locations. Furthermore,
both map-based and route-based sources of spatial information are
likely to be replaced by large scale place or bearing maps as
increased familiarity produces qualitative changes in the processing
and neural representation of spatial information (Jacobs & Schenk,
2003. Further work is needed to determine the range over which
different navigational mechanisms operate (e.g., Beason et al., 1997;
Schmidt-Koenig, 1966, 1970), whether gradient maps provide unicoordinate or bicoordinate information, and the extent to which
olfactory cues are involved in
providing map information as opposed to distinguishing home from non-home,
triggering acquisition and recall of stored map and compass reference information,
and recognizing familiar sites.
[1] Original data from this experiment are no longer available due to
the closing of the Schmidt-Koenig laboratory at the University of T³bingen.
[2]
Given
recent evidence for spatial, temporal, and experiential
heterogeneity of the spatial cues used by pigeons for homing (e.g.,
Beason et al. 1997; Ganzhorn 1990, Wiltschko et al., 1989), nonsense
odor experiments should be carried out using birds of different ages
and levels of experience (see Footnote 3), exposed to different
types of training, and tested at a variety of distances and
directions from the home loft.
[3] In another series of ōnonsense
odorö experiments, young inexperienced birds were exposed to the
control, bottled air, and non-sense odor treatments combined with
anesthetization of the olfactory mucosa at the release site. In
these experiments, only the bottled air group showed significant
homeward orientation when bearings were pooled from four sites
arrayed symmetrically around the release site. Neither controls nor
non-sense odor birds exhibited significant homeward orientation.
For young, inexperienced birds, therefore, the sudden loss of
olfactory input just prior to release, rather than the absence of
olfactory cues per se, appears to have been the cause of
disorientation, again suggesting that non-spatial aspects of
olfactory input can have dramatic effects on homing.
[4] An example of an odor-labeled wind compass would be
the use of
odors associated with the "sea-breeze" to identify the
onshore/offshore directions in a maritime environment.
[5]
In "route-deprivation" experiments, animals are deprived of
potential sources of compass information (magnetic, olfactory,
visual, kinesthetic) during displacement to the testing site to rule
out the use of path integration (also known as "route reversal") to determine
the home direction.
Able, K. P. (1980). Mechanisms of orientation,
navigation and homing. In S. Gauthreaux (Ed.), Animal migration,
orientation and navigation (pp. 283-373). New York: Academic
Press.
Able, K. P. (2000). The concepts and terminology
of bird navigation. Journal of Avian Biology, 32, 174-183.
Able, K.P., & Able, M.A. (1993). Daytime
calibration of magnetic orientation in a migratory bird requires a
view of skylight polarization. Nature, 364, 523-525.
Able, K.P., & Able, M.A. (1997). Development of
sunset orientation cues in a migratory bird: no calibration by the
magnetic field. Animal Behaviour, 53, 363-368.
Baldaccini, N.E., Benvenuti, S., Fiaschi, V., & Papi, F. (1975).
Pigeon navigation: Effects of wind deflection at home cage on homing behaviour.
Journal of Comparative Physiology A.: Neuroethology, Sensory, Neural, and
Behavioral Physiology, 99, 177-186.
Baldocchi, D.D. (1989). Turbulent
transfer in a deciduous forest. Tree Physiology, 5, 357-377.
Beason, R.C., & Semm, P. (1987). Magnetic responses of the
trigeminal nerve system of the Bobolink Dolichonyx oryzivorus.
Neuroscience Letters, 80, 229-234.
Beason, R.C., & Semm, P. (1996). Does the avian ophthalmic nerve
carry magnetic navigational information? Journal of Experimental
Biology, 199, 1241-1244.
Beason, R.C., Wiltschko, R., & Wiltschko, W. (1997). Pigeon homing:
Effects of magnetic pulses on initial orientation. The Auk,
114(3), 405-415.
Benvenuti, S., & Wallraff, H.G. (1985). Pigeon navigation: Site
simulation by means of atmospheric odours. Journal of Comparative
Physiology A, 156, 737-746.
Berthold, P. (1990). Spatiotemporal programs and genetics of
orientation. Experientia, 46, 363-371.
Berthold, P. (1991). Genetic control of migratory behaviour in
birds. TREE, 6, 254-257.
Bingman, V.P., Gagliardo, A., & IoalĶ, P. (1996). Hippocampal
participation in the sun compass orientation of phase-shifted homing
pigeons. Journal of Comparative Physiology A, 179, 695-702.
Bletz,
H., Weindler, P., Wiltschko, R., & Wiltschko, W. (1996). The
magnetic field as reference for the innate migratory direction in
blackcaps (Sylvia atricapilla).
Naturwissenschaften, 83 , 430-432.
Boles, L.C., & Lohmann, K.J. (2003). True navigation and magnetic
maps in spiny lobsters. Nature, 421, 60-63.
Brines, M.L., & Gould, J.L. (1982). Skylight polarization patterns
and animal orientation. Journal of Experimental Biology, 96,
69-91.
Burt,
T., Holland, R., & Guilford, T. (1997). Further
evidence for visual landmarks involvement in the pigeons' familiar
area map. Animal Behavior, 53, 1203-1209.
Clayton, N.S., & Krebs, J.R. (1994). Memory for
spatial and object-specific cues in food-storing and non-storing
birds. Journal of Comparative Physiology A: Sensory Neural and
Behavioral Physiology, 174, 371-379.
Clark, L., & Shah, P.S. (1992). Information content of odor
plumes: What do Leach's Storm Petrels know? In R.L. Doty & D.
Muller-Schwarze (Eds.), Chemical Signals in Vertebrates (Vol.
6, pp. 421-427). New York: Plenum Press.
Courtillot, V., Hulot, G., Alexandrescu, M., le
Mouļl, J.L., & Kirschvink, J.L. (1997). Sensitivity and evolution of
sea-turtle magnetoreception: Observations, modelling and constraints
from geomagnetic secular variation. Terra Nova, 9, 203-207.
Deutschlander, M.E., Borland, S.C., & Phillips
J.B. (1999). Extraocular magnetic compass in newts. Nature, 400,
324-325.
Diego-Rasilla, F.J., Luengo, R.M., & Phillips,
J.B. (2005). Magnetic compass mediates nocturnal homing by the
alpine newt, Triturus alpestris. Behavioral Ecology
and Sociobiology, 58,
361-365.
Emlen, S.T. (1970). Celestial rotation: Its
importance in the development of migratory orientation. Science,
170, 1198-1201.
Emlen, S.T. (1975). Migration: Orientation and
navigation. In D.S. Farner & J.R. King (Eds.), Avian biology
(Vol. 5, pp. 129-219). New York: Academic Press.
Fischer, J.H., Freake, M.J., Borland, S.C., &
Phillips, J.B. (2001). Evidence for the use of a magnetic map by an
amphibian. Animal Behavior, 62, 1-10.
Fischer, J., Munro, U., & Phillips, J.B. (2003).
Magnetic navigation by an avian migrant? In P. Berthold, E. Gwinner,
& E. Sonnenschein (Eds.), Avian migration (pp. 423-432).
Springer-Verlag, Heidelberg-New York.
Fleissner, G., Holtkamp-R÷tzler, E., Hanzlik, M.,
Winklhofer, M., Fleissner, G., Petersen, N., & Wiltschko, W. (2003).
Ultrastructural analysis of a putative magnetoreceptor in the beak
of homing pigeons. Journal of Comparative Neurology, 458,
350-360.
Foa, A., Bagnoli, P., & Giongo, F. (1986). Homing
pigeons subjected to section of the anterior commissure can build up
two olfactory maps in the deflector lofts. Journal of
Comparative Physiology A, 159, 465-472.
Freake, M.J., & Phillips, J.B. (2005).
Light-dependent shift in bullfrog tadpole magnetic compass
orientation: Evidence for a common magnetoreception mechanism
in anuran and urodele amphibians. Ethology, 111, 241-254.
Gagliardo, A., IoalĶ, P., & Bingman, V.P. (1999).
Homing in pigeons: The role of the hippocampal formation in the
representation of landmarks used for navigation. Journal of
Neuroscience, 19, 311-315.
Gagliardo, A., IoalĶ, P., Odetti, F., Kahn, M.C.,
& Bingman, V.P. (2004). Hippocampal lesions do not dirupt
navigational map retention in homing pigeons under conditions when
amp acquisition is hippocampal dependent. Behavioural Brain
Research, 153, 35-42.
Gagliardo, A., Mazzotto, M., & Bingman, V.
(1997). Piriform cortex ablations block navigational map learning in
homing pigeons. Behavioural Brain Research, 86, 143-148.
Ganzhorn, J.U. (1990). Towards the map of the
homing pigeon. Animal Behaviour, 40, 65-78.
Ganzhorn, J.U. (1992). Geographical patterns in
the initial orientation of homing pigeons in upstate New York. Animal
Behavior, 44, 931-941.
Ganzhorn, J.U. (1995). Patterns of air pollution
as model for the physical basis for olfactory navigation in pigeon
homing. Journal of Ornithology, 136, 159-165.
Ganzhorn, J.U., & Schmidt-Koenig, K.
(1990). Initial orientation of homing pigeons around T³bingen. In R.
van der Elzen, K.L. Schuchmann, & K. Schmidt-Koenig (Eds.),
Current topics in avian biology (pp. 305-309). Deutsche
Ornithologen Gesselschaft, Bonn.
Gould, J.L. (1982). The
map sense of pigeons. Nature, 296, 205-211.
Gould, J.L. (1985). Are animal maps magnetic? In
J.L Kirschvink, D.S. Jones, & B.J. McFadden (Eds), Magnetite
biomineralization and magnetoreception in organisms: A new
biomagnetism (pp. 257-268). New York and London: Plenum Press.
Grant, D., Anderson, O., & Twitty, V.
(1968). Homing orientation by olfaction in newts Taricha
rivularis. Science, 160, 1354-1355.
Griffin, D.R. (1952). Bird navigation. Biological
Reviews of the Cambridge Philosophical Society, 27, 359-400.
Guilford, T., Gagliardo, A., Chappell, J.,
Bonadonna, F., Burt, T., & Holland, R. (1998). Homing pigeons use
olfactory cues for navigation in England. Journal of Experimental
Biology, 201, 895-900.
Gwinner, E., Wiltschko, W. (1978). Endogenously
controlled changes in the migratory direction of garden warbler,
Sylvia borin. Journal of Comparative Physiology, 125,
267-273.
Hays, G.C., ┼kesson, S., Broderick, A.C., Glen,
F., Godley, B.J., Papi, F., & Luschi, P. (2003). Island-finding
ability of marine turtles. Proceedings of the Royal Society of
London, Series B, 270 (supplement), S5-S7.
Helbig, A.J. (1992). Ontogenetic stability of
inherited migratory directions in a nocturnal bird migrant:
Comparison between the first and second year of life. Ecology,
Ethology, Evolution, 4, 375-388.
Helbig, A.J. (1996). Genetic basis, mode of
inheritance and evolutionary changes of migratory directions in
paleaarctic warblers (Aves: Sylviidae). Journal of
Experimental Biology, 199(1), 49-55.
Hutchison, B.A., & Hicks, D.D. (Eds.). (1985).
The Forest-atmosphere interaction: Proceedings of the Forest
Environmental Measurements Conference, Oak Ridge, Tennessee. Dordrecht, Holland: D. Reidel.
IoalĶ, P. (1984). Magnets and pigeon
orientation. Monitore Zoologico Italiano (N.S.), 18, 347-358.
IoalĶ, P., Gagliardo, A., & Bingman, V.P. (2000).
Hippocampal participation in navigational map learning in young
homing pigeons is dependent on training experience.
European Journal of Neuroscience, 12, 742-750.
IoalĶ, P., Papi, F., Fiaschi, V., & Baldaccini, N.E. (1978). Pigeon
navigation: effects upon homing behaviour by reversing wind
direction at the loft. Journal of Comparative Physiology, 128,
285-295.
Jacobs, L.F., & Schenk, F. (2003). Unpacking the
cognitive map: The parallel map theory of hippocampal function.
Psychological Review, 110, 285-315.
Joly, P., & Miaud, C. (1989). Fidelity to the
breeding site in the alpine newt Triturus alpestris.Behavioural Processes, 19, 47-56.
Joly, P., & Miaud, C. (1993). How does a
newt find its pond? The role of chemical cues in migrating
newts. Ethology, Ecology, Evolution, 5, 447-455.
Kiepenheuer, J. (1978). Pigeon homing: A repetition of the deflector loft experiment. Behavioral Ecology
and Sociobiology, 3, 393-395.
Kiepenheuer, J. (1979). Pigeon homing:
Deprivation of olfactory information does not affect the deflector
loft effect. Behavioral Ecology and Sociobiology, 6, 11-22.
Kiepenheuer, J. (1982). Pigeon orientation: A
preliminary evaluation of factors involved or not involved in the
deflector loft effect. In F. Papi & H.G.
Wallraff (Eds.), Avian navigation (pp. 203-210). Berlin,
Germany: Springer-Verlag.
Kiepenheuer, J. (1985). Can pigeons be fooled
about the actual release site position by presenting them
information from another site. Behavioral Ecology and
Sociobiology, 18, 75-82.
Lohmann, K.J., & Lohmann, C.M.F. (1996).
Orientation and open-sea navigation in sea turtles. Journal of
Experimental Biology, 199, 73-81.
Lohmann, K.J., Lohmann, C.M.F., Ehrhart, L.M., Bagley, D.A., &
Swing, T. (2004). Animal behaviour: Geomagnetic map used in
sea-turtle navigation . Nature, 428, 909-910.
Matthews, G.V.T. (1963). The orientation of pigeons as affected by
the learning of landmarks and by the distance of displacement. Animal
Behaviour, 11, 310-317.
Moore, B.R. (1980). Is the homing pigeon's mag geomagnetic?
Nature, 285, 69-70.
Moore, F.R. (1987). Sunset and the orientation behavior of migratory
birds. Biological Reviews, 62, 65-86.
Mora,
C.V., Davidson, M., Wild, J.M., & Walker, M.M. (2004).
Magnetoreception and its trigeminal mediation in the homing pigeon.
Nature, 432,508-511.
Mouritsen, H. (2003). Spatiotemporal orientation strategies of
long-distance migrants. In P. Berthold, E. Gwinner,
& E. Sonnenschein (Eds.), Avian Migration (pp.
493-513). Berlin: Springer Verlag.
Muheim, R., Bõckman, J., & ┼kesson, S. (2002). Magnetic compass
orientation in European robins is dependent on both wavelength and
intensity of light. Journal of Experimental Biology, 205(24),
3845-3856.
Muheim, R., Moore, F.R., & Phillips, J.B. (2006a). Calibration of
magnetic and celestial compass cues in migratory birds- a review of
cue-conflict experiments. Journal of Experimental Biology, 209,
2-17.
Muheim, R., Phillips, J.B., & Akesson, S. (2006b). Polarized light
cues underlie compass calibration in migratory songbird. Science,
313, 837-839.
Munro, U., Munro, J.A., & Phillips, J.B. (1997a). Evidence for a
magnetite-based navigational 'map' in birds. Naturwissenschaften,
84, 26-28.
Munro, U., Munro, J.A., Phillips, J.B., & Wiltschko, W. (1997b).
Effect of wavelength of light and pulse magnetization on different magnetoreception systems in a migratory bird. Australian Journal
of Zoology, 45, 189-198.
Nevitt, G.A., Reid, K., & Trathan, P. (2004). Testing olfactory
foraging strategies in an Antarctic seabird assemblage. Journal
of Experimental Biology, 207, 3537-3544.
Papi,
F. (1990). Olfactory navigation in birds. Experientia, 46,
352-362.
Perdeck, A.C. (1958). Two types of orientation in migrating
Starlings Sturnus vulgaris and Chaffinches Fringilla
coelebs as revealed by displacement experiments. Ardea, 46,
1-37.
Phillips, J.B. (1986a). Magnetic compass orientation in the Eastern
red-spotted newt Notophthalmus viridescens. Journal of
Comparative Physiology A, 158, 103-109.
Phillips, J.B. (1986b). Two magnetoreceptor pathways in a migratory
salamander. Science, 233, 765-767.
Phillips, J.B. (1996). Magnetic navigation. Journal of
Theoretical Biology, 180, 309-319.
Phillips, J.B., Adler, K., & Borland, S.C. (1995). True navigation
by an amphibian. Animal Behaviour, 50, 855-858.
Phillips, J.B., & Borland, S.C. (1992a). Magnetic compass
orientation is eliminated under near-infrared light in the Eastern
red-spotted newt, Notophthalmus viridescens. Animal
Behaviour, 44, 796-797.
Phillips, J.B., & Borland, S.C. (1992b). Wavelength-specific effects
of light on magnetic compass orientation of the Eastern red-spotted
newt Notophthalmus viridescens. Ecology, Ethology,
Evolution, 4, 33-42.
Phillips, J.B., & Borland, S.C. (1994). Use
of a specialized magnetoreception system for homing by the Eastern
red-spotted newt, Notophthalmus viridescens. Journal of
Experimental Biology, 188, 275-291.
Phillips, J.B., Borland, S.C., Freake, M.J., Brassart, J., &
Kirschvink, J.L. (2002a). `Fixed-axis' magnetic orientation by
an amphibian: Non-shoreward-directed compass orientation,
misdirected homing or positioning a magnetite-based map detector in
a consistent alignment relative to the magnetic field?
Journal of Experimental Biology, 205(24), 3903-3914.
Phillips, J.B., & Deutschlander, M.E. (1997). Magnetoreception in
terrestrial vertebrates: Implications for possible mechanisms of EMF
interaction with biological systems. In R.G. Stevens, L.E. Andrews,
& B.W. Wilson (Eds.), The melatonin hypothesis: Electric
power and the risk of breast cancer (pp. 111-172). Columbus, OH:
Battelle Press.
Phillips, J.B., Freake, M.J., Fischer, J.H., & Borland, S.C.
(2002b). Behavioural titration of a magnetic map coordinate.
Journal of Comparative Physiology A, 188, 157-160.
Phillips, J.B., & F.R. Moore (1992).
Calibration of the sun‑compass of a migratory bird by sunset
polarized light patterns. Behavioral Ecology & Sociobiology,
31,189‑194.
Phillips, J.B., & Waldvogel, J.A. (1982). Reflected light cues
generate the short-term deflector-loft effect. In F. Papi & E.
Wallraff (Eds.), Avian navigation (pp. 190-202). Berlin, Springer-Verlag.
Phillips, J.B., & Waldvogel, J.A. (1988). Celestial polarized
patterns as a calibration reference for sun compass of homing
pigeons. Journal of Theoretical Biology, 131, 55-67.
Ritz,
T., Adem, S., & Schulten, K. (2000). A model for photoreceptor-based magnetoreception in birds. Biophysical Journal, 78, 707-718.
Ritz,
T., Thalau, P., Phillips, J.B., Wiltschko, R., & Wiltschko, W.
(2004). Resonance effects indicate a radical-pair mechanism for
avian magnetic compass. Nature, 429(6988),
177-180.
Rodda, G.H. (1984). The orientation and
navigation of juvenile alligators: Evidence of magnetic sensitivity.
Journal of Comparative Physiology A, 154, 649-658.
Rodda, G.H., & Phillips, J.B. (1992). Navigational systems develop
along similar lines in amphibians, reptiles and birds. Ecology,
Ethology, Evolution, 4, 43-51.
Saint
Paul, Uv. (1953). Nachweis der sonnenorientierung
bei nõchtlich ziehenden V÷geln. Behaviour, 6, 1-7.
Sauer, E.G.F., & Sauer, E.M. (1960). Star navigation of
nocturnal migrating birds. Cold Spring Harbor Symposium
Quantitative Biology, 25, 463-473.
Schmidt-Koenig, K (1966). Ueber die entfernung als
parameter bei der anfangsorientierung der brieftaube.
Zeitschrift f³r Vergleichende Physiologie, 52, 33-55.
Schmidt-Koenig, K. (1970). Entfernung und
heimkehrverhalten der brieftaube. Zeitschrift f³r
Vergleichende Physiologie, 68, 39-48.
Schmidt-Koenig, K. (1979). Avian
orientation and navigation. London, Academic Press.
Schmidt-Koenig,
K. (1990). The sun compass. Experientia, 46, 336-342.
Sinsch, U. (1990a). Migration and orientation in anuran amphibians. Etholology Ecology & Evolution,
2, 65-79.
Sinsch, U. (1990b).The orientation behaviour
of three toad species (genus Bufo) displaced from the breeding site.
In W. Hanke (Ed.) Biology and physiology of amphibians (Vol. 38, pp.
73-83). G. Fischer Verlag, Fortschritte der Zoologie.
Skiles, D.D. (1985). The geomagnetic field: Its nature, history, and
biological relevance. In J.L. Kirschvink, D.S. Jones, & B.J.
McFadden (Eds.), Magnetite biomineralization and magnetoreception
in organisms: A new biomagnetism (pp. 43-102). New York and
London: Plenum Press.
Walcott, C. (1991). Magnetic maps in pigeons. In P. Berthold
(Ed.) Orientation in birds (pp. 38-51). Basel:
Birkhõuser Verlag.
Walcott, C., & Schmidt-Koenig, K, (1973).
The effect on pigeon homing of anesthesia during
displacement. Auk, 90,
281-286.
Waldvogel, J.A. (1987). Olfactory navigation
in homing pigeons: Are the current models atmospherically
realistic? Auk, 104, 369-379.
Waldvogel, J.A., Benvenuti, S., Keeton, W.T., & Papi, F.
(1978). Homing pigeon orientation influenced by deflected winds at
home loft. Journal of Comparative Physiology, 128, 297-301.
Waldvogel, J.A., & Phillips, J.B. (1982). Pigeon
Homing: New experiments involving permanent-resident deflector-loft
birds. In F. Papi, & H.G. Wallraff (Eds.), Avian
Navigation (pp. 179-189). Berlin: Springer Verlag.
Walker, M., Diebel, C.E., Haugh, C.V., Pankhurst, P.M., Montogmery,
J.C., & Green, C.R. (1997). Structure and function of the
vertebrate magnetic sense. Nature, 390, 371-376.
Walker, M.M., Dennis, T.E., & Kirschvink, J.L. (2002). The
magnetic sense and its use in long-distance navigation by animals.
Current Opinion in Neurobiology, 12, 735-744.
Wallraff, H.G. (1990). Navigation by homing pigeons. Ethology,
Ecology, & Evolution, 2, 81-115.
Wallraff, H.G. (2004). Avian olfactory navigation: Its empirical
foundation and conceptual state. Animal Behaviour, 67,
189-204.
Wallraff, H.G., Kiepenheuer, J., Neumann, M.F., & Streng, A.
(1995). Homing experiments with starlings deprived of the
sense of smell. Condor, 97, 20-26.
Wehner, R. (1998). Navigation in context: Grand theories and basic
mechanisms. Journal of Avian Biology, 29,
370-386.
Weindler, P., Wiltschko, R., & Wiltschko, W. (1996). Magnetic
information affects the stellar orientation of young bird migrants.
Nature, 383, 158-160.
Wiltschko, R. (1996). The function of olfactory input in
pigeon orientation: Does it provide navigational information or play
another role? Journal of Experimental Biology, 199(1),
113-119.
Wiltschko, R., Sch÷ps, M., & Kowalski, U. (1989). Pigeon
homing: Wind exposition determines the importance of olfactory
input.
Naturwissenschaften, 76, 229-231.
Wiltschko, R., & Wiltschko, W. (1995a). Magnetic orientation
in animals. Berlin: Springer-Verlag.
Wiltschko, R., & Wiltschko, W. (1998). Pigeon homing: Effect of
various wavelengths of light during displacement.
Naturwissenschaften, 85, 164-167.
Wiltschko, R., & Wiltschko, W. (2000). A strategy for beginners!
Reply to Wallraff (2000). Animal Behaviour, 60, F37-F43.
Wiltschko, R., & Wiltschko, W. (2003). Avian navigation: From
historical to modern concepts. Animal Behaviour, 65, 257-272.
Wiltschko, W., Daum, P., Fergenbauer-Kimmel, A., & Wiltschko, R.
(1987). The development of the star compass in garden warblers
Sylvia borin. Ethology, 74, 285-292.
Wiltschko, W., Munro, U., Ford, H., & Wiltschko, R. (1993). Red
light disrupts magnetic orientation of migratory birds. Nature,
364, 525-527.
Wiltschko, W., Munro, U., Ford, H., & Wiltschko, R.
(2003). Lateralization of magnetic compass orientation in
silvereyes, Zosterops l. lateralis. Australian Journal of
Zoology, 51, 597-602.
Wiltschko, W., Munro, U., Wiltschko, R., & Kirschvink, J.L. (2002a).
Magnetite-based magnetoreception in birds: The effect of a biasing
field and a pulse on migratory behavior. Journal of Experimental
Biology, 205(19), 3031-3037.
Wiltschko, W., Traudt, J., G³nt³rk³n, O., Prior, H., & Wiltschko, R.
(2002b). Lateralization of magnetic compass orientation in a
migratory bird. Nature, 419, 467-470.
Wiltschko, W., & Wiltschko, R. (1972). Magnetic compass of European
robins. Science, 176, 62-64.
Wiltschko, W., & Wiltschko, R. (1995b). Migratory orientation of
European robins is affected by the wavelength of light as well as by
a magnetic pulse. Journal of Comparative Physiology A, 177,
363-369.
Wiltschko, W., & Wiltschko, R. (1999). The effect of yellow and blue
light on magnetic compass orientation in European robins Erithacus
rubecula. Journal of Comparative Physiology A, 184, 295-299.
Wiltschko, W., & Wiltschko, R. (2001). Light-dependent
magnetoreception in birds: The behaviour of European robins,
Erithacus rubecula, under monochromatic light of various
wavelengths and intensities. Journal of Experimental Biology, 204,
3295-3302.
Yeagley, H.L. (1947). A preliminary study of a physical basis of
bird navigation. Part I. Journal of Applied Physics, 18(12),
1035-1063.
Yeagley, H.L. (1951). A preliminary study of a physical basis of
bird navigation. Part II. Journal of Applied Physics, 22(6),
746-760.
AcknowledgementsSupport was
provided by National Science Foundation grants IBN02-16957
and IBN04 25712 (to JBP), and the Swiss National Science
Foundation (postdoctoral fellowship) to R.M |
®2006 All copyrights for the individual chapters are retained by the
authors. All other material in this book is copyrighted by the
editor, unless noted otherwise. If there has been an error with
regards to unacknowledged copyrighted material, please contact the
editor immediately so that this can be corrected. Permissions for
using material in this book should be sent to the editor.




